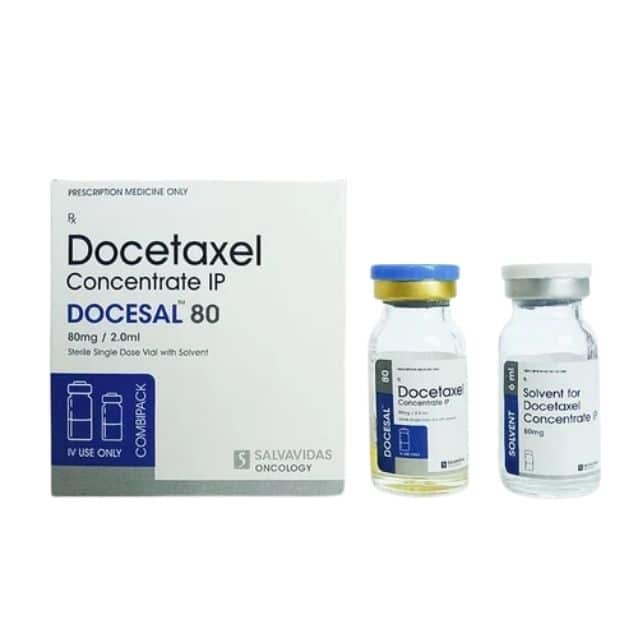
Docetaxel PhaRes Injection – (80mg
11,730.00৳ Original price was: 11,730.00৳ .11,143.00৳ Current price is: 11,143.00৳ .
Docetaxel PhaRes IV Infusion
Docetaxel Trihydrate
80 mg/vial
ZAS Corporation
(Mfg. by: Thymoorgan Pharmazie GmbH, Germany)
| Weight | 0.15 kg |
|---|
Out of stock
0
People watching this product now!
Description
Indications
Docetaxel PhaRes is a microtubule inhibitor indicated for:
- Breast Cancer (BC): single agent for locally advanced or metastatic BC after chemotherapy failure; and with doxorubicin and cyclophosphamide as adjuvant treatment of operable node-positive BC
- Non-small Cell Lung Cancer (NSCLC): single agent for locally advanced or metastatic NSCLC after platinum therapy failure; and with cisplatin for unresectable, locally advanced or metastatic untreated NSCLC
- Castration-Resistant Prostate Cancer (CRPC): with prednisone in metastatic castration-resistant prostate cancer
- Gastric Adenocarcinoma (GC): with cisplatin and fluorouracil for untreated, advanced GC, including the gastroesophageal junction
- Squamous Cell Carcinoma of the Head and Neck (SCCHN): with cisplatin and fluorouracil for induction treatment of locally advanced SCCHN
Pharmacology
Docetaxel is an antineoplastic agent, which acts by disrupting the microtubular network in cells that is essential for vital mitotic and interphase cellular functions. Docetaxel promotes the assembly of tubulin into stable microtubules while simultaneously inhibiting their disassembly. Docetaxel binds to free tubulin thereby decreasing the critical intracellular concentration of tubulin. The promoted polymerization of microtubules leads to the production of microtubule bundles without normal function and to the stabilization of microtubules, resulting in the inhibition of mitosis in cells. The binding of Docetaxel to microtubules does not alter the number of protofilaments in the bound microtubules; in that, it differs from other spindle poisons. Docetaxel was found to be cytotoxic in vitro against various murine and human tumor cell lines, and against freshly excised human tumor cells in clonogenic assays. In addition, Docetaxel was found to be active on a number of cell lines overexpressing the p-glycoprotein, which is encoded by the multidrug resistant gene.
Dosage & Administration
Administer in a facility equipped to manage possible complications (e.g., anaphylaxis). Administer intravenously (IV) over 1 hr every 3 weeks. PVC equipment is not recommended. Use only a 21 gauge needle to withdraw docetaxel from the vial.
- BC locally advanced or metastatic: 60 mg/m2 to 100 mg/m2 single agent
- BC adjuvant: 75 mg/m2 administered 1 hour after doxorubicin 50 mg/m2 and cyclophosphamide 500 mg/m2 every 3 weeks for 6 cycles
- NSCLC: after platinum therapy failure: 75 mg/m2 single agent
- NSCLC: chemotherapy-naive: 75 mg/m2 followed by cisplatin 75 mg/m2
- HRPC: 75 mg/m2 with 5 mg prednisone twice a day continuously
- GC: 75 mg/m2 followed by cisplatin 75 mg/m2 (both on day 1 only) followed by fluorouracil 750 mg/m2 per day as a 24-hr IV (days 1-5), starting at end of cisplatin infusion
- SCCHN: 75 mg/m2 followed by cisplatin 75 mg/m2 IV (day 1), followed by fluorouracil 750 mg/m2 per day as a 24-hr IV (days 1–5), starting at end of cisplatinin fusion; for 4 cycles
- SCCHN: 75 mg/m2 followed by cisplatin 100 mg/m2 IV (day 1), followed by fluorouracil 1000 mg/m2 per day as a 24-hr IV (days 1–4); for 3 cycles
For all patients:
- Premedicate with oral corticosteroids
- Adjust dose as needed
Interaction
Docetaxel PhaRes is a CYP3A4 substrate. In vitro studies have shown that the metabolism of docetaxel may be modified by the concomitant administration of compounds that induce, inhibit, or are metabolized by cytochrome P450 3A4.
Contraindications
Docetaxel is contraindicated in patients with:
- Neutrophil counts of <1500 cells/mm3
- A history of severe hypersensitivity reactions to docetaxel or to other drugs formulated with polysorbate 80. Severe reactions, including anaphylaxis, have occurred
Side Effects
The most common adverse reactions across all docetaxel indications are infections, neutropenia, anemia, febrile neutropenia, hypersensitivity, thrombocytopenia, neuropathy, dysgeusia, dyspnea, constipation, anorexia, nail disorders, fluid retention, asthenia, pain, nausea, diarrhea, vomiting, mucositis, alopecia, skin reactions, and myalgia. Incidence varies depending on the indication. Adverse reactions are described according to indication. Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Pregnancy & Lactation
Based on findings in animal reproduction studies and its mechanism of action, docetaxel can cause fetal harm when administered to a pregnant woman. There is no information regarding the presence of docetaxel in human milk, or on its effects on milk production or the breastfed child. No lactation studies in animals have been conducted.
Precautions & Warnings
- Docetaxel PhaRes should be administered under the supervision of a physician experienced in the use of antineoplastic agents
- There is a higher risk of developing severe adverse reactions including toxic death and fatal gastrointestinal hemorrhage in patients with hepatic impairment
- Docetaxel PhaRes therapy should not be given to patients with neutrophil counts of less than 1,500 cells/mm3
- Fatal cases of enterocolitis, including ischemic colitis, colitis and neutropenic enterocolitis have been reported
- Severe hypersensitivity reactions, with a potential fatal outcome, requiringb immediate discontinuation of Docetaxel PhaRes may occur. Patients should be closely monitored
- Treatment related acute myeloid leukemia may occur. No studies have been conducted to assess the carcinogenic potential of Docetaxel PhaRes
Overdose Effects
There were a few reports of overdosage. There is no known antidote for docetaxel overdose. In case of overdose, the patient should be kept in a specialised unit and vital functions closely monitored. In case of overdosage, exacerbation of adverse events may be expected. The primary anticipated complications of overdosage would consist of bone marrow suppression, peripheral neurotoxicity and mucositis. Patients should receive therapeutic G-CSF as soon as possible after discovery of overdose. Other appropriate symptomatic measures should be taken, as needed.
Therapeutic Class
Cytotoxic Chemotherapy
Storage Conditions
Store between 2°C and 25°C and protected from light. Freezing does not adversely affect the product.
Reviews (0)
Only logged in customers who have purchased this product may leave a review.



Reviews
There are no reviews yet.